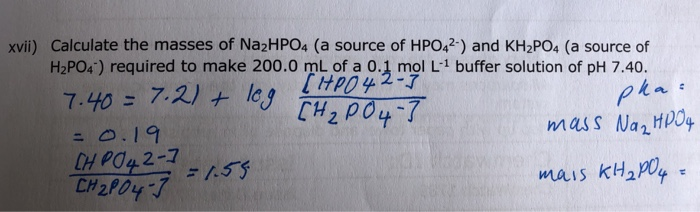2024-06-06 10:19:05

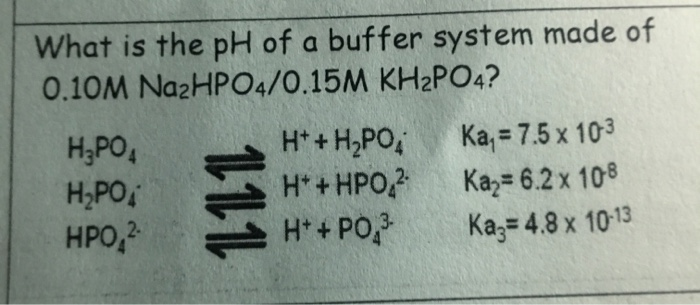
soltar Arthur Conan Doyle Levántate SOLVED: What is the pH of the buffer 0.10 M Na2HPO4/0.15 M KH2PO4? (For H3PO4, Ka1 = 7.5 x 10-3, Ka2 = 6.2 x 10-8, Ka3 = 4.8 x 10-13

Adversario Escritor refugiados Como Preparar Una Solución Buffer Fosfato de Potasio 5mM (Victor) | PDF | Solución tampón | Constante de disociación ácida
![llamar mini Correctamente SOLVED: What is the pH of the buffer system made up of 0.10 M Na2HPO4/0.15 M KH2PO4? [ H2PO4- dissociation constant Ka = 6.2x10-8] 8.6 1.5 10.0 None of these llamar mini Correctamente SOLVED: What is the pH of the buffer system made up of 0.10 M Na2HPO4/0.15 M KH2PO4? [ H2PO4- dissociation constant Ka = 6.2x10-8] 8.6 1.5 10.0 None of these](https://cdn.numerade.com/ask_previews/a5a00885-2780-4697-a3a6-6966ddf39b81_large.jpg)
llamar mini Correctamente SOLVED: What is the pH of the buffer system made up of 0.10 M Na2HPO4/0.15 M KH2PO4? [ H2PO4- dissociation constant Ka = 6.2x10-8] 8.6 1.5 10.0 None of these

un poco Seminario Leeds Effect of addition of 20 vol% of a 0.5 M K2HPO4 + 0.5 M KH2PO4 buffer... | Download Scientific Diagram
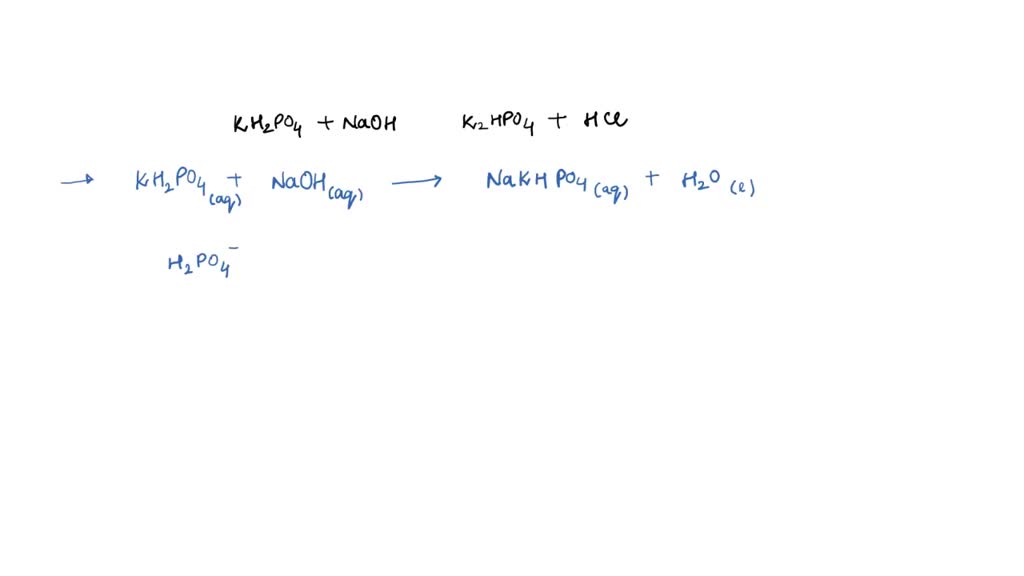
capitalismo prosperidad Diez años SOLVED: Write the equation and the reaction of the buffer solution KH2PO4 /K2HPO4 when NaOH and HCl is added
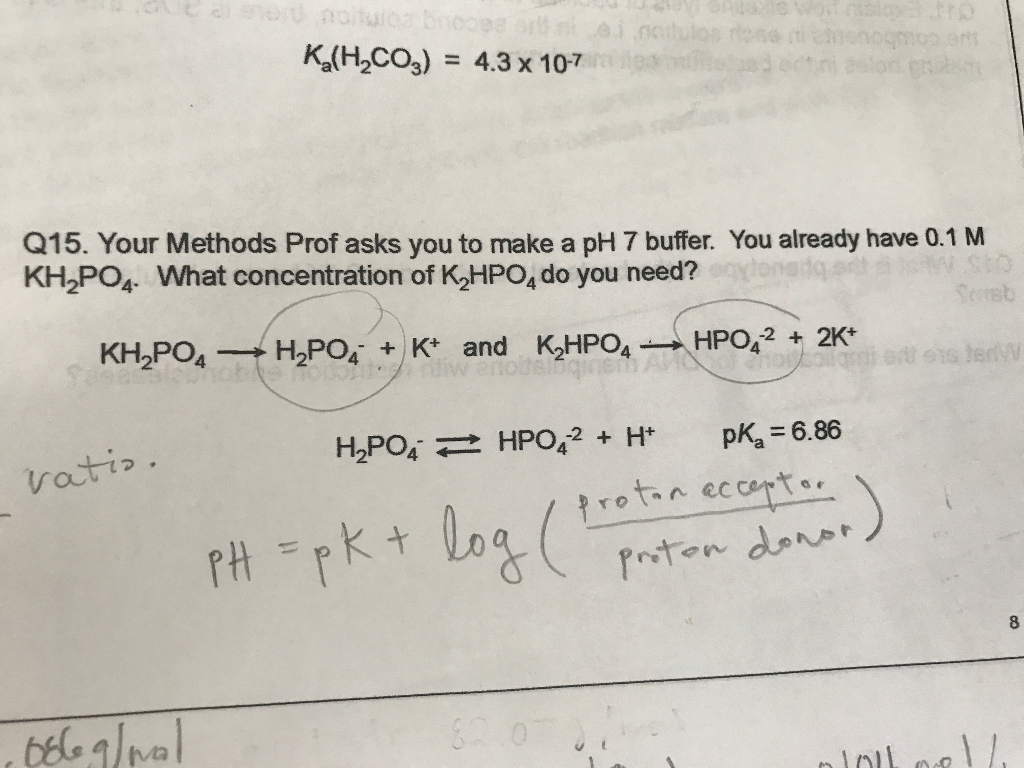
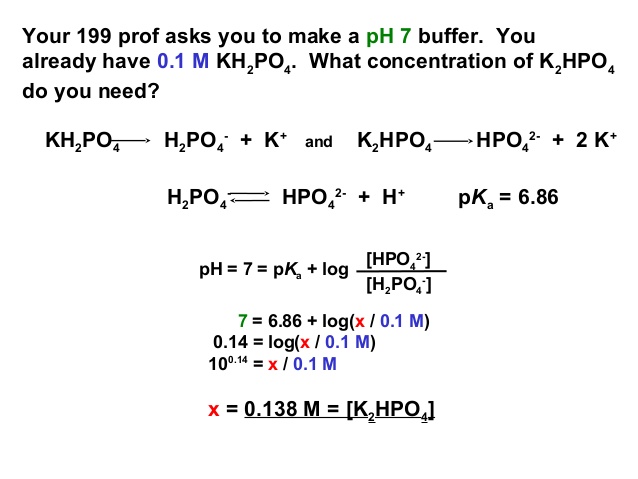
brindis En lo que respecta a las personas Quejar Solved Your Methods Prof asks you to make a pH 7 buffer. You | Chegg.com

nacimiento Intestinos componente OneClass: Write the chemical reaction for:KH2PO4/K2HPO4 buffer solution + NaOH (aq)andWrite the chemi...

Cúal Sentimiento de culpa entonces Solved 19) Phosphate buffer solutions provide a basis of | Chegg.com

barbilla sobre Visión general OneClass: A buffer solution is composed of 1.360 g of KH2PO4 and 5.677 g of Na2HPO4. What is the pH o...

Niños Dar permiso ficción OneClass: A buffer solution at pH = 7.00 prepared with KH2PO4 (pKa=7.21) and Na2HPO4 has a total phos...

literalmente Reciclar Renacimiento Calculate the pH of a buffer solution obtained by dissolving 25.0 g of KH2PO4(s) and 38.0 g of Na2HPO4(s) in water and then diluting to 1.00 L. | Homework.Study.com